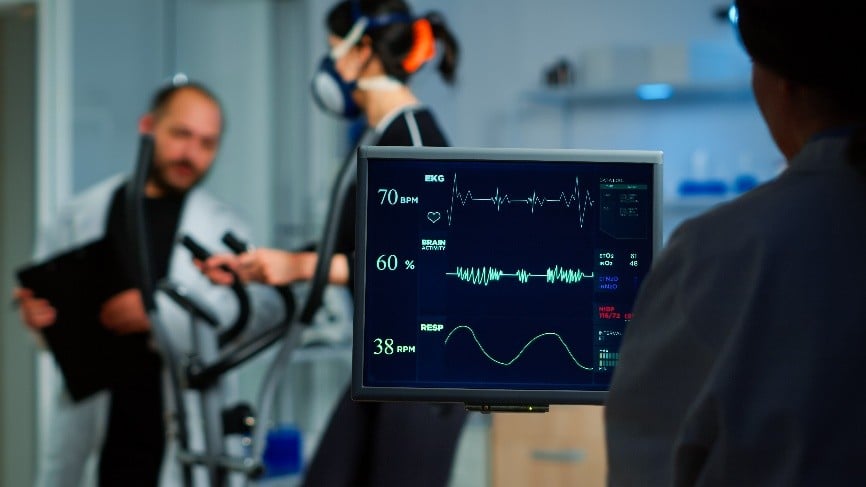
Measuring Power, Force and Strength following ACL surgery using ForceDecks technology – a clinical study

Dr Alex Kumar
Consultant in Pain Medicine
- 29 March, 2023
- knee pain
- Podiatry
- 5 min read
We have teamed up with Queen Mary University of London’s Sports Medicine programme to investigate ACL reconstruction surgery and it results 9 months + post-surgery.
The study will involve a 1-hour testing session using effective data collection tools such as ForceDecks to test your current strength levels, balance, and more. This kind of testing allows us to have measurable data to see how successful the surgery and subsequent rehab treatment has been for you and your ACL recovery.
The session will take place at one of two Pure Sports Medicine locations, depending on where is most convenient for you.
If you’d like to take part, click the button below.
What is the purpose of the study and what would taking part involve?
Purpose: We want to assess the strength, power, and force that your legs can produce following ACL surgery and compare these values to people who have not. This will allow us to understand how people recover after surgery and how we can improve the care that we provide.
To do this, we would need you to participate in a single testing day. During testing we will ask you to complete different tasks. The tasks are progressively more challenging dependant on the time since your surgery.
Test
Static Mid thigh pull
Strength Hamstring, Quadriceps, Glutes and Adductors
Double leg jumps
Single leg jumps
Double leg drop jump from a 40cm box
Single leg drop jump from a 40cm box
SL hop, Triple hop
Crossover hop test
Testing will take around an hour.
If you accept the invitation to join the project, you will be asked screening questions, we will take some baseline data, such as height, weight etc. You will be asked to fill in a simple form to capture some information on your sport, the level of participation you compete in and general symptoms.
The results from the testing day will then be reviewed and written up to complete your participation in the research project. You will be asked to complete two questionnaires regarding your overall knee symptoms. Once we have received your questionnaires and the all the results have been evaluated you will be provided with a post testing evaluation report.
Why am I being invited?
The researchers are inviting you to participate in this research study because you are over 18 and have had ACL reconstructive surgery at least 9 months ago.
You should not take part in this study if you have any other current lower limb injuries, have had a revision of your previous ACL surgery on the same leg or have any neurological issues.
Do I have to take part?
There is no requirement to take part in the study, the information sheet has been written to provide you with the information to make that decision. If you decide to take part, you can withdraw at any time. De-identified data collected on you up to the time of withdrawal will be used in the overall analysis.
What are the possible benefits of taking part?
The benefit of taking part is the assessment on the force decks that unless in a professional setting many do not have any access to. The testing report will provide you with information on your current strength, power, and force levels that you can use to help guide further gym or rehabilitation sessions.
What are the possible disadvantages and risks of taking part?
There are some risks associated with study participation. Mainly, focusing on potential injury during one of the hops or jumps by a fall or over-stretching. To decrease the likelihood of risk you will be provided with a warm-up from a specialist physiotherapist and evaluated pre-session. If you have any problems during the testing day and would not like to continue, you will be able to withdraw.
Expenses and payments
Travel expenses will be covered for individuals who participate in the study if they can provide researchers with a receipt to expense.
What information about me will you be collecting?
Once participants join the study, they will be asked to initial and sign a consent form and then asked to fill in a form that will ask for: age, sport, gender, time since return to play and weight.
Researchers will then provide participants with forms that will capture your activity level, knee function and pain levels. During the testing we will be collecting information on your strength, power, and force as you move on the force decks.
How will my data be stored and who will have access to it?
Your data will be stored in a de-identified format. We will provide you with an ID number which will replace your name. Any data that is collected from yourself will be only identified through this ID number.
A single form that links your name to the ID number will be stored on the QMUL (Queen Mary University of London) cloud storage system until the study is completed and will then be destroyed. The de-identified data will be stored in the QMUL Sports and Exercise Medicine Department. Records containing personal information will not be accessible to anyone outside of the research team.
When and how will my data be destroyed?
Data provided from the hops and jumps after the study has been completed and written up will be handled by the university and destroyed after a period of 5 years. This is in line with the Queen Mary University records retention schedule.
How will my data be used and shared?
The data that will be collected during the study will be used for a dissertation project, this will be initially presented to colleagues of the QMUL Sports and Exercise Medicine Department. It will subsequently be written in preparation for publication within a sports medicine journal.
Under what legal basis are you collecting this information?
Queen Mary University of London processes personal data for research purposes in accordance with the lawful basis of ‘public task.’
Please read Queen Mary’s privacy notice for research participants containing important information about your personal data and your rights in this respect. If you have any questions relating to data protection, please contact Queen Mary’s Data Protection Officer, Queens’ Building, Mile End Road, London, E1 4NS or [email protected] or 020 7882 7596.
What will happen if I want to withdraw from this study?
There are no issues if a participant wants to withdraw during the research project. Also, during the testing day, if there are any problems that arise, individuals can withdraw if they want to.
What should I do if I have any concerns about this study?
If you have any concerns about the manner in which the study was conducted, in the first instance, please contact the researcher(s) responsible for the study Dr Simon Lack, [email protected]. If you have a complaint which you feel you cannot discuss with the researchers, then you should contact the Research Ethics Facilitators by e-mail: [email protected]. When contacting the Research Ethics Facilitators, please provide details of the study title, description of the study and QMERC (Queen Mary Ethics of Research Committee) reference number (where possible), the researcher(s) involved, and details of the complaint you wish to make.
Additional Information
- Version number and date: Version number 1.0 20/12/2021
- Researcher’s name: Dr Simon Lack and team
- Queen Mary Ethics of Research Committee reference number: QMREC2018/48/088

Advice
Over the last 20+ years our experts have helped more than 100,000 patients, but we don’t stop there. We also like to share our knowledge and insight to help people lead healthier lives, and here you will find our extensive library of advice on a variety of topics to help you do the same.
OUR ADVICE HUBS See all Advice Hubs